The osteoarthritis (OA) microenvironment causes transplanted stem cells to rapidly lose viability and undergo aberrant differentiation, presenting a major obstacle to stem cell transplantation (SCT) therapy.
On October 9, 2025, Professor Qianjun He from the School of Materials Science and Engineering and Hydrogen Science Center at Shanghai Jiao Tong University, Researcher Bo Yu from Tongji Hospital Affiliated with Tongji Medical College of Huazhong University of Science and Technology, Chief Physician Qining Yang from Jinhua Central Hospital, and others published a research paper entitled "Local sustained H₂ release assisted stem cell transplantation for enhanced osteoarthritic cartilage regeneration" in Cell Biomaterials, a subjournal of Cell.
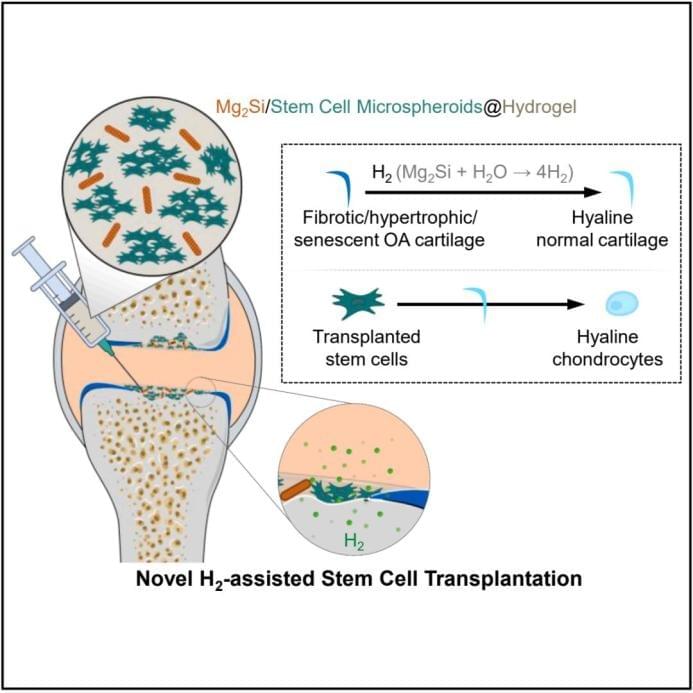
The study found that hydrogen gas (H₂) can reverse the chondrocyte phenotype in osteoarthritis (OA). Furthermore, the team developed Mg₂Si nanosheets (MSN) and co-encapsulated them with adipose-derived stem cell microspheres in a hydrogel, enabling ultra-durable hydrolytic hydrogen production (up to 28 days). This strategy of locally sustained H₂ release-assisted stem cell transplantation effectively promotes cartilage regeneration in osteoarthritis.

Stem cell transplantation (SCT) is an advanced technology in regenerative medicine that promotes the repair of damaged tissues by transplanting stem cells to compensate for their lost regenerative capacity. Based on stem cells' extensive expansion potential, multidirectional differentiation plasticity, low immunogenicity, and diverse sources, SCT has achieved remarkable progress, including 27 clinically approved stem cell therapeutic products and more than 800 ongoing clinical trials utilizing stem cells for treatment.
However, SCT faces a major challenge: once transplanted into a hostile pathological microenvironment, stem cells tend to rapidly lose viability and undergo aberrant differentiation. Remodeling the hostile pathological microenvironment into a stem cell-favorable physiological microenvironment is particularly critical for improving the therapeutic efficacy of SCT, yet it remains challenging.
Chondrocytes are the key cells in articular cartilage, responsible for secreting cartilage matrix and paracrine factors, inducing stem cell differentiation into chondrocytes, and maintaining cartilage homeostasis. In the osteoarthritis (OA) microenvironment, this homeostasis is disrupted by chronic inflammation and oxidative stress, which drive the differentiation of hyaline chondrocytes into fibrotic, hypertrophic, and senescent phenotypes, thereby leading to cartilage degeneration and OA progression—a process that is clinically difficult to halt or reverse. The hostile OA microenvironment causes transplanted stem cells to rapidly lose viability and reduces their hyaline chondrogenic differentiation potential.
Therefore, effectively remodeling the hostile OA microenvironment, particularly eliminating inflammation and restoring OA chondrocytes to a hyaline phenotype, is crucial for improving the efficacy of stem cell therapy in OA chondrogenesis, yet it remains a formidable challenge.
Owing to its strong ability to scavenge hydroxyl radicals (·OH), hydrogen gas (H₂) exhibits broad-spectrum anti-oxidative stress, anti-inflammatory, anti-apoptotic, and anti-aging effects. At concentrations below its saturation level (0.8 mmol), it possesses high biosafety for normal cells/tissues with no obvious toxic or side effects. Moreover, it demonstrates excellent permeability into cartilage/bone tissue, showing great potential for assisting OA cartilage repair and regeneration.
It has been reported that H₂ can prevent hydrogen peroxide (H₂O₂)-induced depletion of the hyaline phenotype and viability of hyaline chondrocytes in vitro via its antioxidant effects; however, whether H₂ can reverse the chondrocyte phenotype in the OA microenvironment remains unclear. Furthermore, high-concentration H₂ delivery at specific sites is essential for its effective OA-modulating function. Hydrolytic hydrogen-producing hydrogels or scaffold implantation represent an ideal approach for local high-concentration H₂ delivery, but the hydrogen production duration of existing hydrolytic hydrogen-generating materials does not exceed 7 days, which fails to meet the time window required for bone/cartilage repair.
In this latest study, the research team discovered that the loss of the hyaline chondrocyte phenotype in the OA microenvironment can be restored by H₂ targeting mitochondrial dysfunction. Accordingly, the team developed size-tunable magnesium silicide (Mg₂Si) nanosheets (MSN) with controllable hydrolytic hydrogen production behavior. By co-encapsulating MSN with adipose-derived stem cell microspheres in a gelatin methacryloyl (GelMA)/hyaluronic acid methacrylate (HAMA) hydrogel, a hydrogel implant capable of sustained H₂ release (up to 28 days) was constructed.
This implant remodels the OA microenvironment by inhibiting inflammation and cartilage degeneration, significantly improving the survival rate of transplanted stem cells in vivo and maintaining their hyaline chondrogenic differentiation properties, thereby enhancing the efficacy of stem cells in repairing critical-sized cartilage defects in OA. This strategy of locally sustained H₂ release to remodel the hostile pathological microenvironment will open a new avenue for improving the outcomes of cell transplantation.
Highlights of the study:
- Discovery that H₂ can reverse the chondrocyte phenotype in osteoarthritis;
- Development of Mg₂Si nanosheets (MSN) capable of ultra-durable hydrolytic hydrogen production;
- Local sustained H₂ supply can assist stem cell transplantation.
Original Aritcle:
